Hepatitis C Blog
Greg Jefferys Hepatitis C blog deals with all the issues associated with hepatitis C
Sofosbuvir and Sovaldi: A brief History.
- September 30, 2017
- By Greg Jefferys
- 1 Comment
Hepatitis C Treatment: A Short History of Sofosbuvir and Sovaldi.
It is now two and a half years since generic Sovaldi was released in India, in May 2015, at a price that was 100th of the cost of Gilead’s branded Sovaldi in the USA.
Here is a brief history of Sofosbuvir and Sovaldi.
Sovaldi is the brand name for Sofosbuvir and the Sovaldi pill contains 400 mg of Sofosbuvir. Sovaldi was approved in the USA in December 2013 and was a breakthrough treatment for Hepatitis C sufferers offering higher cure rates than any other available treatment at that time without the terrible side effects that came with the Interferon based treatments.

Michael Sofia: The man who invented Sofosbuvir
Sofosbuvir was invented in 2007 by, and named after, Michael Sofia, a scientist working for a company called Pharmasset. Sofosbuvir was first tested on humans in 2010 with amazing results and Gilead purchased Pharmasset in 2011.
Gilead did not spend any money researching Sofosbuvir, it purchased a product that was ready to go.
Further testing resulted in Sofosbuvir being approved by the FDA in December 2013 and in the first quarter of 2014 Gilead sold over US$2 Billion worth of Sovaldi. By the end of 2014 sales of Sovaldi had exceeded US$10 billion.
Given that one 12 week treatment of Sovaldi costs less than US$100 to manufacture and it was sold for US$84,000 or almost one thousand times the cost of manufacture, this was a very profitable year for Gilead.
Whilst the huge profits were great news for Gilead and its shareholders the exorbitant price was bad news for most of the more than 100 million people around the world with Hepatitis C because the price put Sovaldi beyond the reach of most people, even in wealthy countries like the USA.
However it was not all smooth sailing for Gilead. The Indian Patents Office did not consider Sofosbuvir sufficiently innovative to be granted a patent and immediately India’s very capable pharmaceutical manufacturers saw a huge opportunity to make generic versions of Sovaldi.
Here Gilead acted fast.
Gilead knew that Sofosbuvir could be made easily and cheaply and it did not want international markets flooded with cheap and effective versions of Sovaldi so it entered into negotiations with the major Indian pharmaceutical companies and in exchange for giving the Indian pharmaceutical companies a license that included all the intellectual property associated with Sofosbuvir the Indians would pay Gilead a royalty on all sales and would agree not to sell into any of Gilead’s major markets, such as the USA and Europe.
This agreement meant that Gilead could keep making huge profits from the wealthier countries and it would still get some income from the poorer countries, which it was not selling into anyway.
In May 2015 the first licensed generic Sofosbuvir became available from India at a retail price that was about 100th the price in the USA and other western countries.
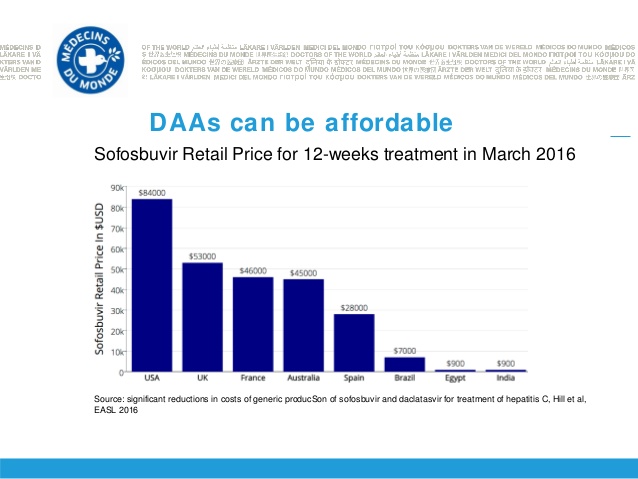
Prices for Sofosbuvir around the world differ greatly even though the drug is identical
And here began an interesting thing.
Most people with Hep C in Western countries like the USA who did not have high quality health insurance could not afford the $84,000 cost of Sovaldi.
Conversely most people in Third World countries could not afford the 0 cost of generic Sovaldi.
So people from the west began, by various means, to purchase generic Sovaldi. This meant that the India pharmaceutical manufacturers could produce higher volumes of Sofosbuvir, thus bringing the price down and making generic Sovaldi cheaper in Third World countries.
Of course Gilead tried to stop the flow of generics into its Western markets, mostly by fear campaigns, warning of fake, even toxic, generics however this campaign was only partially successful and as more and more people were treated with generic Sovaldi doctor, nurses and people with Hep C all saw the results. The fact was that treating Hepatitis C with generic Sofosbuvir gave exactly the same cure rates but at 100th the price.
The rest, as they say, is history.

Sofosbuvir and Sovaldi should be available to all people with Hepatitis C at affordable prices
- Post Categories
- Uncategorized
Greg Jefferys
Tags
- Sofosbuvir And Daclatasvir
- Sofosbuvir And Ledipasvir
- Sofosbuvir And Ribavirin
- Sofosbuvir And Velpatasvir
- Sofosbuvir Canada Price
- Sofosbuvir Cure Rate
- Sofosbuvir In India Price
- Sofosbuvir Price
- Sofosbuvir Where To Buy
- Sofosbuvir With Daclatasvir
- Sovaldi And Daklinza
- Sovaldi Canada Price
- Sovaldi Cost
- Sovaldi Daklinza Genotype 3
- Sovaldi In Pakistan
- Sovaldi India
- Sovaldi Indonesia
- Sovaldi Or Harvoni
- Sovaldi Price
1 Comment
Leave a Comment Cancel Reply
Join my Hep C Support Group.
Talk privately to other people
with Hep C in a closed group.
Disclaimer
Greg Jefferys’ blog is provided for informational purposes and is not intended as Medical advice, diagnosis, or treatment.
Whilst Greg Jefferys is doing a PhD it is not in medicine. Any advice offered is offered in good faith and based on an extensive general knowledge of Hepatitis C and access to generic Hepatitis medicines Greg Jefferys has acquired through his work as an advocate and activist
The Hep C Buyers Club is not a company or corporate entity but simply a loose structure intended to offer a free information to people with Hepatitis C
Other Books
Click here for other books by Greg Jefferys.
for Kindle
I have converted this diary into a kindle book for folk who might like it in that format. I have added a lot more depth than the original diary contains, it’s more of a complete story in book format. I have priced it as low as Kindle allows me to @ 99 cents. If you are interested just click here to go to the Kindle page.
Recent Posts
-
Hepatitis C Treatment and Liver Cancer June 19, 2024
-
Motivate C a Profitable Hep C Initiative April 29, 2024
-
After Hep C Healthy Liver Diet September 4, 2023
-
Fear of Hep C Treatment April 14, 2023
-
My Letter to Joe Biden March 2, 2023
-
Dormant Hepatitis C November 27, 2022
-
How Much water to drink during Hep C treatment October 13, 2022
-
Hep C and Peripheral Neuropathy August 21, 2022
-
Fatigue Brain Fog and Hepatitis C August 18, 2022
-
Hep C and Liver Cancer May 18, 2022
Contact Us
If you have any questions please reach out by email, or complete the below form.
Greg Jefferys3439 Channel Highway, Woodbridge, Tasmania, 7161.
Email: gregjefferys@outlook.com



Dear Admin,
I have used Sovaldi medicine a couple of years ago and I am very much satisfied about result. Thanks for manufacturing.
Now, I am suffering from haemorrhoids (Piles). My question is, if your company is producing any medicine for piles then please be very kind and suggest me. I will appreciate your response as I am having a great confidence in your medicines.
Best regards,
Shoaib